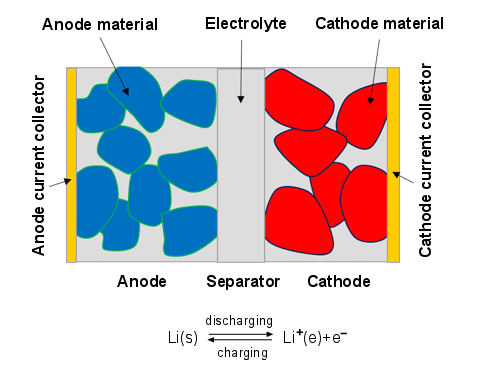
Lithium-ion battery is typically made by stacking/rolling-up many layers of the electrodes to form a sandwich-like structure. Each electrode consists of the anode current collector, anode, separator, cathode, and cathode current collector as shown in Figure 18.1: Schematic structure of an electrode pair.
During discharge, lithium stored in the anode material diffuses out, undergoes electrochemical reaction at the solid-electrolyte interface (SEI), and dissolves in the electrolyte phase as lithium-ion species. Then lithium-ion species diffuses further from the anode side to the cathode side. As the lithium-ion species undergoes electrochemical reaction at the cathode SEI, lithium is intercalated into the cathode material. During the charge process, the lithium and lithium-ion move in the opposite direction. For information about using the lithium-ion battery model, see Simulating the Lithium-ion Battery in the Fluent User's Guide.
For additional information, see the following sections:
The detailed lithium-ion battery model simulates the detailed physics occurring in the charging and discharging processes from first principles. The model is derived from the concentrated solution theory [474], [212]. The governing equations are the mass and charge balance equations expressed as:
where
|
|
|
|
|
|
The mass and electric current flux can be computed as:
where
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
is defined by:
(18–12) |
where
|
|
|
|
|
|
The electrochemical reaction occurs at the SEIs. The reaction rate is determined by the Butler-Volmer equation:
(18–13) |
A linearized form can also be used:
(18–14) |
with
(18–15) |
In Equation 18–13 through Equation 18–15, the following notation is used:
|
|
|
|
|
|
|
|
At the SEIs, although potential and lithium are not continuous, the mass and electric current fluxes must be continuous satisfying the following conditions:
(18–16) |
(18–17) |
Substituting the flux equations (Equation 18–10 and Equation 18–11) into the mass and current conservation equations (Equation 18–8 and Equation 18–8) yields the following:
Mass conservation law:
Charge conservation law:
Ansys Fluent solves Equation 18–18 through Equation 18–21 together with the SEI conditions (Equation 18–16 and Equation 18–17 to obtain the electric and species fields.
Modeling Assumptions
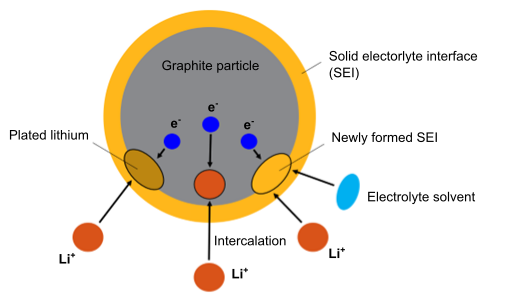
In Ansys Fluent, the battery aging model considers the aging mechanisms of the growth of the solid electrolyte interphase (SEI), the decomposition of metallic lithium (Li plating) on the anode material [724], and the film formation on the cathode. Figure 18.2: Electrochemical Reactions Considered on the Anode in the Aging Model shows the electrochemical reactions that occur on the anode surface during battery charging.
The following electrochemical reactions are considered on the anode:
Intercalation reaction:
(18–22)
This is a desired reaction where
ions intercalate into the graphite.
SEI formation reaction:
(18–23)
In this reaction, the
ions react with the electrolyte solvent to form the SEI film. The SEI is considered to consist of lithium ethylene dicarbonate
, which is formed by the reaction of ethylene carbonate (EC)
with
ions.
Lithium deposition reaction:
(18–24)
In this reaction, the
ions deposit onto the graphite surface, where they partly become metallic lithium (Li-plating) and partly create SEI.
Reactions 2 and 3 are unwanted side reactions that compete with the intercalation reaction and are included in the battery aging model. Note that these two side reactions are prominent and modeled only in the anode during battery charging.
The film layer formed on the cathode particles is called the solid permeable interphase (SPI). The SPI is formed on the electrode particles from reactions with electrolyte components, and unlike the formation of the SEI layer, it is assumed that it is formed during both charging and discharging.
The SEI growth, Li plating, and cathode film growth cause battery aging primarily due to the following effects. Firstly, some of the lithium inventory is lost to form the side reactions, which leads to a decrease in the battery theoretical capacity. Secondly, the surface film generated from the side reactions increases the electric contact resistance in the reactions.
Model Formulation
To model the side reactions, the aging model solves the following equations:
In the above equations, the following notation is used:
As the governing equations have no spatial terms, their evolution depends solely on the local source terms, specifically the local current fluxes of the side reactions. These source terms are formulated by:
(18–28) |
(18–29) |
(18–30) |
In Equation 18–28, the current flux is a function of the
reaction rate constant , the surface concentration of the ethylene dicarbonate (EC)
, the cathodic charge transfer coefficient
, and the reaction overpotential
. The surface concentration of EC,
, is further determined by the diffusive flux of EC across the
surface film as:
(18–31) |
In Equation 18–29, the li-plating flux is a function of
the reaction exchange current density , which is modelled as a constant, the cathodic charge transfer
coefficient
, and the reaction overpotential
.
In Equation 18–30, the cathode film growth reaction rate
is a function of the reaction rate constant , which is modelled as a constant, the surface concentration of
the ethylene dicarbonate (EC)
, and the electrode lithium concentration
. Similar to Equation 18–31, the surface
concentration of EC,
, is also determined by the diffusive flux of EC across the
surface film:
(18–32) |
As a result of a side reaction (SEI formation or lithium deposition), the overpotential of the side reaction is computed as:
(18–33) |
The physical properties of the surface film are functions of the SEI and plated lithium concentrations computed as:
Thickness
:
Electric resistance
(note that only the SEI layer has resistance):
In Equation 18–28 through Equation 18–37, the following notation is used: